|
Because Y removes protons at a pH greater than the pH of neutral water (7), it is considered a base. To describe how a buffer solution (either acidic or basic) can resist. Henderson-Hasselbalch Equation For Acid Buffer. Henderson-Hasselbalch Equation pH pKa + logA-1/HA -log(3.5x10-4) + log0.50/0.80 3.46 + (-0.40) 3.26 BACK TO MAIN PAGE. The principal objectives of the Henderson Hasselbalch equation include the following: To calculate the pH, pOH, H3O+ tot, OH- tot, H3O+ water, and OH- water in a solution containing a strong acid (base) given the initial concentration of the acid (base). 
For example, if you have a base Y with a pKa of 13, it will accept protons and form YH, but when the pH exceeds 13, YH will be deprotonated and become Y. pOH pKb + logSALT/BASE Henderson-Hasselbalch Equation for Base Buffer Use Henderson-Hasselbalch Equation to determine pH for 0.80M HF/0.50M NaF. Even a chemical ordinarily considered a base can have a pKa value because the terms "acids" and "bases" simply refer to whether a species will give up protons (acid) or remove them (base).On the other hand, the pKa value is constant for each type of molecule.It can be used for pH calculation of a solution containing pair of acid and. For example, concentrated vinegar (acetic acid, which is a weak acid) could have a lower pH than a dilute solution of hydrochloric acid (a strong acid). This is so called Henderson-Hasselbalch equation (or a buffer equation). Use the Henderson-Hasselbalch equation to calculate the pI. This is important because it means a weak acid could actually have a lower pH than a diluted strong acid. Calculate the pH of a weak acid solution of known concentration. pH depends on the concentration of the solution.The lower the pKa, the stronger the acid and the greater its ability to donate protons. 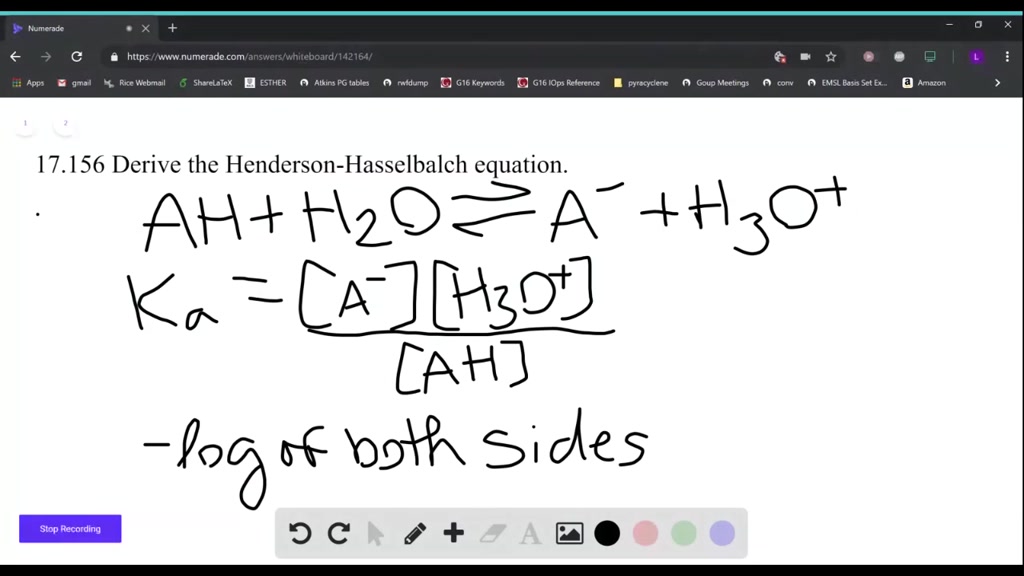
The lower the pH, the higher the concentration of hydrogen ions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
